Quality excellence at the heart of our commitment
The quality department ensures that products and processes comply with regulatory, normative, and customer requirements, from order receipt to product shipment. It also monitors customer complaints, manages non-conformities, conducts audits, and drives continuous improvement of the quality management system.

Quality commitment and ISO certifications
Since 1994, Interprox has been ISO 9001 certified, initially for the manufacturing of proximity detectors, and later for the production of mechatronic assemblies. In 2016, ml&s Suisse SA initiated an additional quality approach aimed at ISO 13485 certification, first for the production of electronic boards, and then for mechatronic systems intended for the medical sector.
Our ISO 13485 certification covers production activities but excludes development and sterilization phases. We apply risk analyses in compliance with ISO 14971, as well as project management procedures based on the APQP (Advanced Product Quality Planning) approach.
Since 2024, Interprox SA and ml&s Suisse SA share a common ISO 9001 certification, enhancing the consistency and rigor of our quality system.
In August 2024, ml&s Suisse SA also received the renewal of its ISO 13485 certification, reaffirming our ongoing commitment to the regulatory requirements of the medical sector.

Supplier and customer quality - a shared commitment
Our quality department supports customers throughout the entire product lifecycle, from the development phase to series production. It also plays an active role with our suppliers, fostering a sustainable partnership and co-building quality together.
In the case of non-conformity, the quality department manages customer and supplier complaints in accordance with the requirements of ISO 9001 and ISO 13485 standards. It handles the receipt, analysis, and follow-up of complaints, identifying root causes using the 6M and 8D methodologies. Corrective and preventive actions are then implemented in collaboration with the relevant departments to prevent recurrence.
This approach ensures:
- Traceability of communications
- Compliance with regulatory requirements
- Satisfaction of stakeholders
- Continuous improvement of product and service quality
Depending on our customers' needs, we can also provide PPAP (Production Part Approval Process) dossiers or PSW (Part Submission Warrant), ensuring full compliance with industry standards.
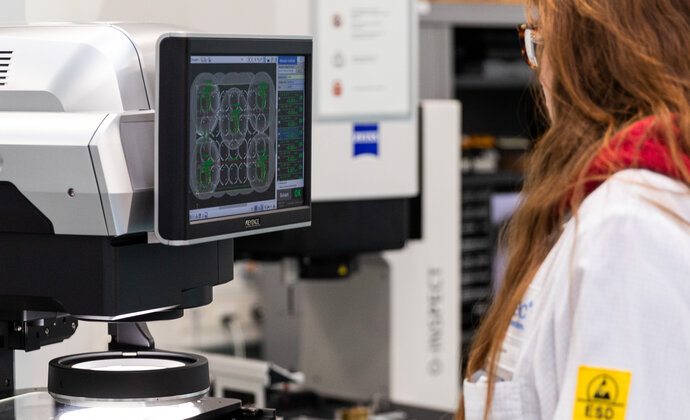
Quality laboratory and product qualification
Our quality department ensures incoming inspection of parts and components using high-precision measurement equipment, guaranteeing compliance with technical and regulatory requirements, particularly according to ISO 9001 and ISO 13485 standards.
We use advanced technologies such as:
- ZEISS O-Inspect 3D CMM (contact and optical measurement)
- KEYENCE optical measurement system
Critical dimensions are verified upon receipt to prevent any defects during production.
This key step helps to:
- Secure the quality of supplies
- Ensure batch traceability
- Strengthen the reliability of the manufacturing process
As part of product qualification, specific measurements are taken in collaboration with internal or external experts.
Our quality laboratory is equipped with environmental chambers for conducting environmental tests, including:
- Temperature cycles
- Hot-humid atmosphere
- Thermal shock tests
- Sterilization tests